the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
CarboCell G/C provides sustained high local bone antibiotic levels with minimal systemic exposure, supporting its therapeutic potential in orthopedic infection management
Nicole Lind Henriksen
Andrea René Jørgensen
Michal Poborsky
Christoph Crocoll
Niranjan G. Kotla
Catrine Jyde Berthelsen
Louise Kruse Jensen
Anders Elias Hansen
Jonas Rosager Henriksen
Introduction: Achieving sustained local antibiotic concentrations is critical for managing orthopedic infections. This proof-of-concept study evaluated release kinetics, local/systemic concentrations, and histological responses to CarboCell G/C, an injectable gentamicin- and clindamycin-eluting depot in a porcine bone void model. Methods: CarboCell G/C containing 111 mg g−1 gentamicin-docusate and 70 mg g−1 clindamycin was injected into and around a tibial bone void in 11 pigs. Antibiotic concentrations in target tissues and systemic spillover were measured at 1–12 h by microdialysis and liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) and in tissue homogenates at 1, 3, and 7 d. Release was quantified by high-performance liquid chromatography (HPLC), and tolerability was assessed histologically. Results: Microdialysis showed high stable antibiotic tissue levels during the first 12 h with minimal systemic spillover. Peak gentamicin and clindamycin concentrations in bone were 730 ± 170 and 3700 ± 700 µg g−1 and 42 ± 15 and 360 ± 120 µg g−1 in soft tissue, respectively. Tissue half-lives ranged from 43–62 h for clindamycin and 40–46 h for gentamicin. Recovered depots had released ∼ 30 %–50 % of their antibiotic content at day 1 and ∼ 60 %–80 % by day 7. Depots elicited a mild–moderate local inflammatory response. Conclusion: CarboCell G/C provides sustained high local antibiotics for at least 7 d with minimal systemic spillover, supporting further evaluation in orthopedic infection management.
- Article
(7449 KB) - Full-text XML
- BibTeX
- EndNote
Orthopedic infections are complex and challenging to treat for several reasons, two of which are the presence of bacterial biofilms and dormant persister cells (Zimmerli et al., 2012). To address these challenges, local antibiotic delivery technologies have gained increasing interest (Jennings et al., 2026). Compared to systemic antibiotic therapy, these technologies offer several potential advantages, including (i) achieving high concentrations of antibiotics in infected regions, (ii) minimizing the risk of antimicrobial resistance, (iii) reducing systemic spillover and associated adverse effects, and (iv) enabling the incorporation of tissue-regenerative biomaterials (Steadman et al., 2023; Jennings et al., 2026). Several antibiotic-eluting biomaterials are available, including collagen, polymethylmethacrylate (PMMA), hydroxyapatite, and calcium sulfate, where cement and void filler technologies are the most commonly described (Steadman et al., 2023). However, concerns about their release kinetics have been raised. For example, for antibiotic-eluting PMMA bone cement, a consensus statement by experts in musculoskeletal infection highlights the risk of early release followed by prolonged subtherapeutic levels of antibiotics (Schwarz et al., 2021). In a systematic review, the total in vitro release of different antibiotics from PMMA bone cement has been reported to be below 10 % even after up to 8 weeks (Martínez-Moreno et al., 2017). In contrast, hydroxyapatite and calcium sulfate biomaterials demonstrate rapid early release of antibiotics (total release > 95 % within 24 h in vitro) (Bezstarosti et al., 2024). Antibiotic delivery technologies with improved and fully characterized release kinetics are therefore warranted. Additionally, data describing antibiotic tissue penetration from such technologies are limited, often restricted to measurements of antibiotic release from explanted biomaterials (Minelli et al., 2004) or measurements of antibiotic concentrations in wound exudate (Fleiter et al., 2014; Colding-Rasmussen et al., 2018). However, such measurements may not necessarily reflect target tissue concentrations, which are central to therapeutic activity (Fink et al., 2011; Steadman et al., 2023). Few studies have reported on concentrations of antibiotics in tissue (Fink et al., 2011; Klinder et al., 2019). Generating data on antibiotic tissue distribution after local delivery is essential to determine whether therapeutic concentrations are achieved and maintained at the target site and guide their clinical use (Müller et al., 2004).
Building on our prior results demonstrating that CarboCell G/C, a dual gentamicin–clindamycin antibiotic-eluting depot technology, can successfully eradicate implant-associated osteomyelitis in a porcine model without systemic antibiotics (Henriksen et al., 2024), this proof-of-concept study aims to characterize the pharmacokinetics and local tolerability. Specifically, we investigated local and systemic concentrations of gentamicin and clindamycin following administration of CarboCell G/C into a bone void.
2.1 CarboCell G/C formulation
CarboCell was formulated as previously described (Henriksen et al., 2024). A total of 111 mg gentamicin-docusate and 70 mg desalted clindamycin were dissolved per gram CarboCell. In this study, 2.5 mL of CarboCell G/C, comprising 42 mg gentamicin and 158 mg clindamycin, was injected into each leg of the pigs.
2.2 Animal studies
Given the exploratory pharmacokinetic nature of the study, sample size was determined based on feasibility and consistency with previous large-animal pharmacokinetic investigations.
2.2.1 Microdialysis (1–12 h)
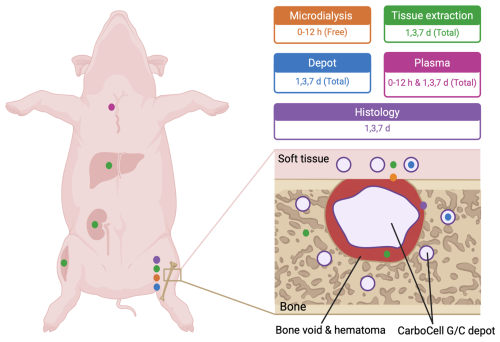
Two 2- to 3-month-old Landrace female pigs weighing 34 and 35 kg were sedated with tiletamine–zolazepam (Virbac, France), ketamine (MSD Animal Health, USA), butorphanol (Orion Pharma, Finland), and xylazine (ScanVet Animal Health, Denmark) and anesthetized with propofol (B.Braun, Germany) and fentanyl (Fresenium Kabi, Germany). A hole was drilled into both proximal tibial bones and expanded with a bone curette to create bone voids of ∼ 1 cm3. CarboCell G/C was injected into the cavity (1 mL), surrounding trabecular bone (1 mL), and soft tissue (0.5 mL), and the wound was closed in three layers (periosteum, soft tissue, skin). A type 70 microdialysis catheter with a 20 mm membrane and 20 kDa cut-off (M Dialysis AB, Stockholm, Sweden, and 107 microdialysis pump (M Dialysis AB), perfused with 0.9 % NaCl at a flow rate of 1 µL min−1) was placed in the bone void–soft tissue interface. The method has been described in detail (Jørgensen et al., 2024). CarboCell G/C injection was time zero. Dialysates were collected initially every 30 min and every 60 min from after 2 h. Blood samples were drawn every hour from 60–720 min, and plasma was collected (3000 × g, 10 min, 5 °C). Catheters were calibrated by retrodialysis (1 mg mL−1 clindamycin and 1 mg mL−1 gentamicin). Pigs were euthanized using pentobarbital (Scanvet). Plasma and dialysates were stored (−80 °C) until liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) analysis.
2.2.2 Tissue samples (1, 3, and 7 d)
Nine 2- to 3-month-old SPF Landrace female pigs (30–35 kg) were subjected to the protocol as described above; however, the procedure was only conducted in the right tibia. Animals received perioperative analgesia (meloxicam PO, Boehringer–Ingelheim) up to 7 d and were monitored in accordance with institutional animal welfare guidelines. Pigs were randomly selected, sedated, and euthanized after 1, 3, or 7 d (n= 3 per timepoint). Blood was drawn prior to surgery and euthanasia, after which it was centrifuged, and plasma was collected. During necropsy, the surgical wound was opened, and soft tissue samples above the bone void were collected. The tibial bone was sagittally sectioned through the void. From one half, samples of the void hematoma and bone tissue below the bone void surface were collected. The other half was placed in 10 % neutral buffered formalin. Kidney, muscle, and liver tissue samples were also collected. Samples were stored (−80 °C) until LC-MS/MS analysis. CarboCell depots were collected for high-performance liquid chromatography (HPLC) analysis of antibiotic release (Fig. 1).
2.2.3 Antibiotic release in mice
Antibiotic release over 21 d in mice was included as a secondary endpoint and performed as previously described (Henriksen et al., 2024). Briefly, two CarboCell G/C depots (50 µL) were injected subcutaneously into the back of 15 anesthetized female, 8- to 9-week-old Balb/C mice (Taconic). After 1, 3, 7, 14, and 21 d, three mice were euthanized by cervical dislocation, and depots were collected.
2.3 LC-MS/MS
2.3.1 Sample preparation
Samples were prepared as previously described (Mikkelsen et al., 2025). Frozen tissue samples were ground in liquid nitrogen. Weighed tissue sample (50–100 mg) were placed in 2 mL low-bind reaction tubes, and extraction was done in three steps. The first extraction was done by adding 500 µL 50 % acetonitrile in Milli-Q water () containing 0.1 % formic acid () and 500 ng mL−1 leucine–enkephaline as internal standards; the mixture was then vortexed and incubated for 20 min in a shaker at 25 °C at 1000 rpm. Samples were vortexed and centrifuged for 10 min at 10 °C and 21 000 ×g. The supernatant was transferred to a new tube. The second extraction was performed with 500 µL 50 % methanol in Milli-Q water () containing 0.1 % formic acid () and 500 ng mL−1 leucine–enkephaline as internal standards, and the third was performed with the same solvent as for the first extraction. Both were followed by the same steps for vortexing, shaking, and centrifugation. Extracts were pooled and diluted 20× with 0.1 % formic acid in water () and filtered through 0.22 µm filter plates prior to LC-MS/MS.
2.3.2 LC-MS/MS
LC-MS/MS was performed as previously described (Mikkelsen et al., 2025). The limit of quantification (LOQ) was 1 ng mL−1 for gentamicin and 0.6 ng mL−1 for clindamycin. Leucine–enkephalin was used as an internal standard for injection control and correction of inter-injection variation. The nominal LOQ for gentamicin and clindamycin in tissues was 0.6 and 0.4 µg g−1, respectively, whereas microdialysis and plasma samples had a nominal LOQ of 0.01–0.02 µg mL−1 for gentamicin and 0.006–0.01 µg mL−1 for clindamycin. Values below the LOQ were not included.
2.4 HPLC
CarboCell G/C depots were analyzed by HPLC, as previously described with the following modifications (Henriksen et al., 2024). The solvent system consisted of mobile phase A (5 % MeCN, 20 mM HFBA in water) and mobile phase B (20 mM HFBA in MeCN) with a gradient of 0 % B for 3 min, 0 %–50 % B over 4 min, 50 %–65 % B for 9 min, 65 %–100 % B for 30 s, 100 % B for 2 min, 100 %–0 % B for 30 s, and 0 % B for 4.5 min. Standards of 1000, 500, 250, 125, 62.5, and 31.25 µg mL−1 gentamicin and clindamycin and 10, 5, 2.5, 1.25, and 0.625 mg mL−1 SuBen were included.
2.5 Histology
Tissue sections (4–5 µm) of formalin-fixed, decalcified, and paraffin-embedded bone were stained with H&E and Masson's Trichrome. Histological evaluation was performed using predefined semi-quantitative criteria. The evaluating pathologist was aware of the experimental condition but blinded to the sampling time point. Within the bone area with surgery-related changes, defined as the distance from the bone void border to the normal trabecular bone and marrow, the morphological tissue response to CarboCell G/C was described, including categorization of inflammatory response (mild, moderate, or severe). Furthermore, fibroplasia (percentage of collagen in 10 × 20 fields translated to a scale of 0–5) and osteoclast counts (maximum 10 cells counted in 10 HPFs) were scored as previously described (Henriksen et al., 2024).
2.6 Data analysis
Pharmacokinetic data were generated in GraphPad Prism version 10 (GraphPad Software, USA). Pharmacokinetic and release data are presented as means ± SEM, and histology data are presented as means ± SD.
Bone void was created in the proximal tibia, followed by injection of CarboCell G/C into the bone void and adjacent bone and soft tissue. Antibiotic concentrations were measured at the bone–soft tissue interface (orange); in bone, soft tissue, liver, muscle, kidney, and void hematoma at days 1, 3, and 7 (green); and in plasma (pink). Antibiotic release from depots recovered from bone and soft tissue was assessed at days 1, 3, and 7 (blue). Bone samples were collected at days 1, 3, and 7 for histological evaluation (purple).
3.1 Tissue antibiotic measurements
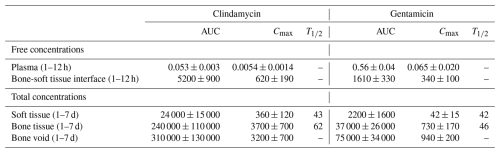
Microdialysis at the bone void–soft tissue interface revealed high initial free antibiotic concentrations, peaking at 340 ± 100 µg mL−1 for gentamicin and 620 ± 190 µg mL−1 for clindamycin, followed by a gradual decline to ∼ 100 and 450 µg mL−1, respectively (Fig. 2A). From day 1 to day 7, total tissue concentrations remained elevated. Gentamicin and clindamycin reached maximum levels on day 1 in bone (730 ± 170 and 3700 ± 700 µg g−1) and soft tissue (42 ± 15 and 360 ± 120 µg g−1) and on day 3 in void hematoma (940 ± 200 and 3200 ± 700 µg g−1) (Table 1). Bone and soft tissue exhibited half-lives of 42–46 h for gentamicin and 43–62 h for clindamycin. By day 7, concentrations remained above 79 ± 19 µg g−1 gentamicin and 710 ± 140 µg g−1 clindamycin in bone (Fig. 2B) and 3.6 ± 1.2 and 37 ± 10 µg g−1 in soft tissue (Fig. 2C). Bone void concentrations were stable throughout, exceeding 290 ± 110 µg g−1 gentamicin and 1500 ± 500 µg g−1 clindamycin (Fig. 2D). Clindamycin, administered at a 3.8-fold higher dose, yielded 3.2-fold greater free drug exposure (AUC1–12 h ratio) and 4.1-, 6.5-, and 10.9-fold higher total exposure in bone void, bone, and soft tissue, respectively (AUC1–7 d ratios).
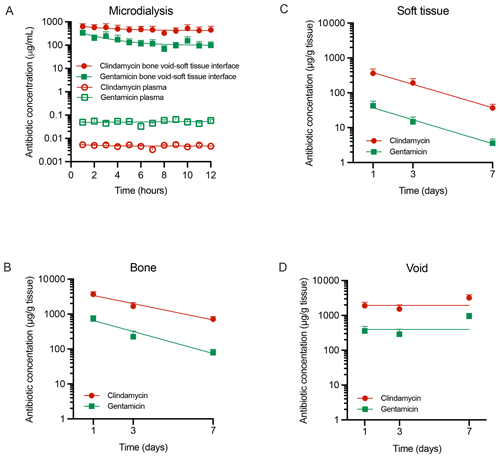
Figure 2Antibiotic tissue concentrations. Concentrations of free clindamycin and gentamicin in (A) plasma and at the bone void–soft tissue interface and total concentrations in samples of (B) bone tissue, (C) soft tissue, and (D) bone void hematoma over 1–7 d. Data are presented as means ± SEM. Microdialysis bone–soft tissue interface: n= 4, microdialysis plasma: n= 2, soft tissue: n= 9–12 (3 values < LOQ for gentamicin day 7), bone tissue: n= 14–15 (1 value < LOQ for gentamicin day 7), and void hematoma: n= 7–9. LOQ: limit of quantification.
3.2 Systemic antibiotic measurements
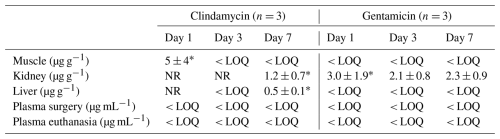
Plasma concentrations remained low and stable during the first 12 h (gentamicin: 0.033–0.065 µg mL−1; clindamycin: 0.0032–0.0054 µg mL−1) (Fig. 2A). Despite a 3.8-fold higher dose, clindamycin exposure was 10-fold lower than gentamicin (AUC1–12 h, Table 1). Beyond 24 h, plasma levels were below the LOQ. The highest tissue concentrations were detected in the kidney (gentamicin: 3.0 ± 1.9 µg g−1; clindamycin: 1.2 ± 0.7 µg g−1), while clindamycin was the only drug quantifiable in muscle (5 ± 4 µg g−1) and liver (0.5 ± 0.1 µg g−1) (Table A1).
3.3 Antibiotic release from depots
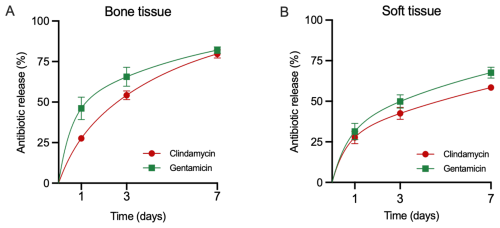
Antibiotic release from CarboCell G/C in pigs demonstrated an initial phase of faster release on day 1, with 46 ± 7 % of gentamicin and 28 ± 2 % of clindamycin released in bone and 31 ± 5 % of gentamicin and 28 ± 4 % of clindamycin released in soft tissue, followed by a slower release over the remaining study period (Fig. 3). By day 7, cumulative release in bone reached 82 ± 2 % for gentamicin and 80 ± 2 % for clindamycin (Fig. 3A), while in soft tissue, release was slightly lower at 68 ± 3 % for gentamicin and 58 ± 1 % for clindamycin (Fig. 3B). Continued release beyond day 7 was confirmed in mice. These data should be interpreted solely as qualitative support rather than as a directly comparable pharmacokinetic model (Fig. A1).
3.4 Morphological bone response
CarboCell G/C depots were present within the bone void and surrounding tissue. Within the void, CarboCell G/C was intermingled with varying amounts of bone debris, fibrin, red blood cells, mononuclear cells, and neutrophils (Fig. 4A). The tissue response was not evaluated on day 1 due to the difficulty in reliably distinguishing bone marrow cells from an early inflammatory response. On day 3, the reaction to CarboCell G/C was mild, consisting of mononuclear cells, such as macrophages, and fibroblasts (Fig. 4B). On day 7, the response varied, with some depots closest to the bone void having a moderate infiltration of mainly fibroblasts and mononuclear cells but also some neutrophils, while others were associated with a mild inflammatory response of similar cells (Fig. 4C–D). The fibrosis score (day 3: 0.47 ± 0.15, day 7: 3.3 ± 0.67), osteoclast number (day 3: 0.13 ± 0.15, day 7: 1.6 ± 1.4), and presence of osteoid increased over time.
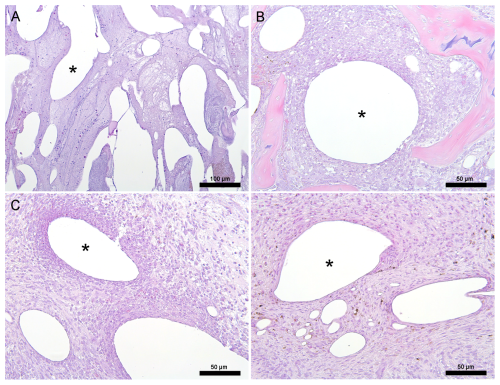
Figure 4Histology. Representative images from pigs euthanized at days 1, 3, and 7, depicting the histological tissue response to CarboCell G/C depots. (A) Day 1: inflammatory response with mononuclear cells and neutrophils throughout the bone void, (B) day 3: mild localized response with fibroblasts and mononuclear cells in bone adjacent to the bone void, (C–D) day 7: response varying from a mild localized response (D) to a heavier localized mononuclear and neutrophilic inflammatory response (C) with more fibroplasia in bone adjacent to the bone void. Images (C) and (D) are from the same animal. * Examples of CarboCell G/C depots. Scale bars correspond to 100 µm in (A) and 50 µm in (B)–(D).
This study demonstrates that local delivery of gentamicin and clindamycin using CarboCell G/C maintains high antibiotic levels in the targeted bone void for at least 7 d, and recovered depots released approximately ∼ 30 %–50 % of their antibiotic content within the first day, reaching ∼ 60 %–80 % cumulative release by day 7. Prior evidence shows that CarboCell G/C effectively eradicates infection in a porcine model of implant-associated osteomyelitis (Henriksen et al., 2024), where systemic antibiotics (Vittrup et al., 2023) and a commercially available gentamicin-eluting hydroxyapatite and calcium sulfate biomaterial (Blirup-Plum et al., 2020) have failed. The present study further confirms that CarboCell G/C delivers therapeutic levels of antibiotics to the infection site, with negligible antibiotic spillover to plasma and off-target tissues, e.g. gentamicin levels remaining below the nephrotoxic limit throughout (Selby et al., 2009). We also observed that CarboCell G/C depots elicited a mild–moderate inflammatory response in bone tissue. Depots with a more pronounced cellular response were located at the bone–void interface, likely reflecting the inflammatory response to surgery. These findings align with previous observations in infected tissue after CarboCell G/C treatment (Henriksen et al., 2024; Fuglsang-Madsen et al., 2024).
The pharmacokinetic data for CarboCell G/C demonstrate clear advantages and robustness over other antibiotic administration forms relevant to orthopedic infections. For example, intravenous gentamicin in pigs resulted in an AUC0–6 h of 26 µg mL−1 h and Cmax of 6.9 µg mL−1 in cancellous bone (Stolle, 2004). Despite the 5.7-fold lower dose, CarboCell G/C resulted in markedly higher bone exposure while maintaining low systemic levels, highlighting its efficiency for local and targeted delivery. Similarly, intraosseous administration of 500 mg diluted vancomycin in pigs exhibited a half-life of 4.6 h in cancellous bone, while CarboCell G/C yielded prolonged half-lives of 46–62 h and markedly higher exposure when quantified over 7 d (CarboCell G/C AUC1–7 d: 37 000 µg g−1 h, intraosseous AUC0–12 h: 6244 µg mL ⋅ h−1), despite providing a 3–12 times lower antibiotic payload (Olsen Kipp et al., 2021). Further, our findings build upon a recent study demonstrating that intraosseous injection of CarboCell G/C can achieve high cancellous bone concentrations over at least 12 h with minimal systemic spillover (Mikkelsen et al., 2025). The concentrations measured in target tissue exceeded typical MIC values for common orthopedic pathogens such as S. aureus by several orders of magnitude, supporting the likelihood of effective antimicrobial activity at the target site. Unfortunately, comparable in vivo pharmacokinetic data for other carrier systems, including PMMA and hydroxyapatite and calcium sulfate-based carriers, are currently lacking.
Notably, in this study, clindamycin showed a 10-fold lower systemic exposure than gentamicin, despite being administered at a 3.8-fold higher dose. This may partly be explained by clindamycin's higher protein binding, which limits the free, systemically available fraction (Lee et al., 2013). Overall, lower antibiotic concentrations were observed in soft tissue compared to the bone void and adjacent cancellous bone, likely due to the smaller CarboCell G/C volume administered. This may reflect a balance between ongoing antibiotic release from surrounding CarboCell G/C depots and local washout kinetics, potentially combined with fluid dynamics that preferentially transport antibiotics toward the void space (Schwarz et al., 2021). Higher antibiotic levels in soft tissue could potentially have been achieved by placing multiple CarboCell G/C depots across a broader area.
By combining in vivo release profiles with tissue concentration data, this study clarifies the relationship between drug release, tissue exposure, and diffusion characteristics. Many reports on antibiotic-eluting technologies claim sustained release based on simple in vitro assays, where depots are placed in buffer and time above the minimum inhibitory concentration (MIC) is measured. Such assays fail to reflect physiological washout kinetics. Reported durations above MIC may instead result from assay-specific factors like buffer volume, depot size, or media exchange timing.
We recognize that this study has several limitations. The antibiotic concentrations were assessed using two quantification methods and interpreted accordingly. Microdialysis samples the free, pharmacologically active fraction of a drug in the extracellular compartment (Azeredo et al., 2014), whereas tissue extraction quantifies total drug concentrations without distinguishing between intra- and extracellular compartments or free and protein-bound fractions (Mouton et al., 2007). The tissue extraction method was optimized by performing triple extractions to ensure accurate quantification of total antibiotic levels for clindamycin and gentamicin (Russo et al., 2023). Gentamicin's low protein binding compared to clindamycin likely affects distribution and detection across sampling methods. Microdialysis may better represent gentamicin's tissue exposure, while for clindamycin it reflects only a fraction of total levels. This aligns with previous findings, showing that gentamicin pharmacokinetics in dialysates closely match bone sample data (Stolle, 2004). Moreover, since the extracellular compartment accounts for 98 %–99 % of bone tissue volume, dilution from intracellular contents during homogenization is minimal, supporting the fact that measured bone concentrations closely approximate extracellular levels (Pea, 2009). Also, the antibiotic concentrations were measured in healthy tissue, which may differ from infected tissue (Zhao et al., 2019). Finally, given that CarboCell G/C is biodegradable, longer-term studies on antibiotic delivery and biocompatibility are warranted.
In conclusion, CarboCell G/C achieved high, sustained local concentrations of clindamycin and gentamicin for at least 7 d with minimal systemic exposure. This technology may improve treatment efficacy while reducing reliance on systemic antibiotics and the risk of antimicrobial resistance.
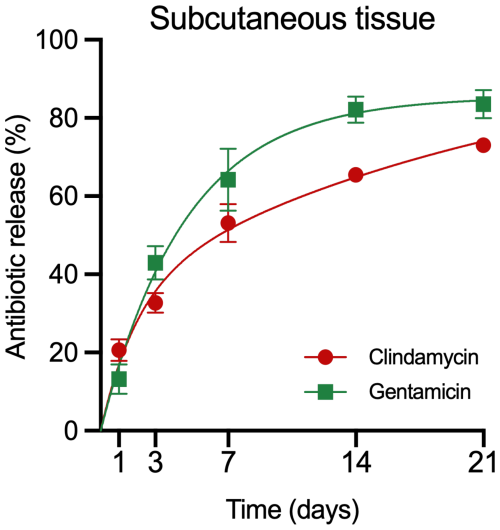
Figure A1Antibiotic release from depots in mice. Accumulated release of gentamicin and clindamycin from CarboCell G/C depots in subcutaneous tissue at 1, 3, 7, 14, and 21 d after administration in mice. Data are presented as means and SEM. n= 5–6 depots at each timepoint (Balb/C mice, female, 8 weeks old (Janvier labs France), acclimatized at least 7 d before commencing study procedures, ad lib access to water and food, housed and monitored according to guidelines from the Danish Animal Inspectorate).
Data are available from the corresponding author.
Conceptualization: AEH, JRH, NLH; methodology: CC, MP, JRH; formal analysis: CC, MP, NK, NLH, resources: AEH, AJ, CB, JRH, NLH, MB; writing – original draft: AEH, JRH , NLH; writing – review and editing: all authors; visualization: NLH; project administration: AEH, JRH; funding acquisition: AEH, JRH, NLH.
Anders E. Hansen and Jonas R. Henriksen are co-inventors on patents covering the CarboCell G/C technology. The peer-review process was guided by an independent editor, and the authors have no other competing interests to declare.
Animal studies were approved by the Danish Animal Experiments Inspectorate (2022-15-0201-01314, 2020-15-0201-00713) and complied with ARRIVE guidelines and the University Institutional Board. Animals were obtained from an approved commercial breeder and housed in nationally approved animal research facilities under controlled enriched environmental conditions. Animals were acclimatized for > 7 d prior to experimentation and monitored daily by veterinary staff.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
The authors thank Kasper With Nielsen and Dennis Brok for technical assistance. AI-assisted language tools were used to support editorial revisions.
This research has been supported by the Innovationsfonden (grant no. 4294-00008B) and the Novo Nordisk Fonden (grant no. NNF21OC0072770).
This paper was edited by Parham Sendi and reviewed by two anonymous referees.
Azeredo, F. J., Dalla Costa, T., and Derendorf, H.: Role of microdialysis in pharmacokinetics and pharmacodynamics: current status and future directions. Clin. Pharmacokinet., 53, 205–212, https://doi.org/10.1007/s40262-014-0131-8, 2014.
Bezstarosti, H., Van Lieshout, E. M. M., Van Den Hurk, M. J. B., Kortram, K., Oprel, P., Koch, B. C. P., Croughs, P. D., and Verhofstad, M. H. J.: In vitro elution of gentamicin from CERAMENT® G has an antimicrobial effect on bacteria with various levels of gentamicin resistance found in fracture-related infection, Clin. Orthop. Relat. R., 482, 885–891, http://https://doi.org/10.1097/CORR.0000000000002975, 2024.
Blirup-Plum, S. A., Bjarnsholt, T., Jensen, H. E., Kragh, K. N., Aalbæk, B., Gottlieb, H., Bue, M., and Jensen, L. K.: Pathological and microbiological impact of a gentamicin-loaded biocomposite following limited or extensive debridement in a porcine model of osteomyelitis, Bone Joint Res., 9, 394–401, https://doi.org/10.1302/2046-3758.97.BJR-2020-0007.R1, 2020.
Colding-Rasmussen, T., Horstmann, P., Petersen, M. M., and Hettwer, W.: Antibiotic Elution Characteristics and Pharmacokinetics of Gentamicin and Vancomycin from a Mineral Antibiotic Carrier: An in vivo Evaluation of 32 Clinical Cases, J. Bone Joint Infect., 3, 234–240, https://doi.org/10.7150/jbji.26301, 2018.
Fink, B., Vogt, S., Reinsch, M., and Büchner, H.: Sufficient release of antibiotic by a spacer 6 weeks after implantation in two-stage revision of infected hip prostheses, Clin. Orthop. Relat. R., 469, 3141–3147, https://doi.org/10.1007/s11999-011-1937-4, 2011.
Fleiter, N., Walter, G., Bösebeck, H., Vogt, S., Büchner, H., Hirschberger, W., and Hoffman, A.: Clinical use and safety of a novel gentamicin-releasing resorbable bone graft substitute in the treatment of osteomyelitis/osteitis, Bone Joint Res., 3, 223–229, https://doi.org/10.1302/2046-3758.37.2000301, 2014.
Fuglsang-Madsen, A. J., Henriksen, N. L., Chávez, E. S., Kvich, L. A., Birch, J. K. M., Hartmann, K. T., Eriksen, T., Bjarnsholt, T., Gottlieb, H., Andresen, T. L., Jensen, L. K., Henriksen, J. R., and Hansen, A. E.: Eradication of Staphylococcus aureus in implant-associated osteomyelitis by an injectable in situ-forming depot antibiotics delivery system, J. Infect Dis., 230, 614–623, https://doi.org/10.1093/infdis/jiae139, 2024.
Henriksen, N. L., Serrano-Chávez, E., Fuglsang-Madsen, A., Jensen, L. K., Gottlieb, H., Bue, M., Andresen, T. L., Henriksen, J. R., and Hansen, A. E.: Gentamicin and clindamycin antibiotic-eluting depot technology eradicates S. aureus in an implant-associated osteomyelitis pig model without systemic antibiotics, Antimicrob. Agents Ch., 68, e00691-24, https://doi.org/10.1128/aac.00691-24, 2024.
Jennings, J. A., Abdelbary, H., Abdulla, F. S., Arafah, O., Benzouak, T., Choe, H., Coenye, T., Coraça-Huber, D. C., Drago, L., Gustavo Garcia, R., Goh, G. S., Hamilton, J., Hamoudeh, R., Hickok, N. J., Jensen, L. K., Lee, H. G., Li, B., Manzary, M., Markovics, A., McDonald, K., Fintan Moriarty, T., Muthukrishnan, G., Nishitani, K., Norton, N. J., Oral, E., Parvizi, J., Del Pozo, J., Priddy, L. B., Raafat, D., Saeed, K., Spiegel, C., Schwarz, E. M., Siverino, C., Trobos, M., Tubbs, A., and Yeasmin, R.: 2025 International Consensus Meeting on Musculoskeletal Infection: summary from biofilm workgroup on treatment of biofilm-related infection and preclinical models, J. Orthop. Res., 44, e70169, http://https://doi.org/10.1002/jor.70169, 2026.
Jørgensen, A. R., Hansen, J., Bue, M., Hanberg, P., and Stilling, M.: Microdialysis as a sampling tool for the chemotherapeutic agent doxorubicin, J. Pharm. Biomed. Anal., 239, 115872, https://doi.org/10.1016/j.jpba.2023.115872, 2024.
Klinder, A., Zaatreh, S., Ellenrieder, M., Redanz, S., Podbielski, A., Reichel, T., Bösebeck, H., Mittelmeier, W., and Bader, R.: Antibiotics release from cement spacers used for two-stage treatment of implant-associated infections after total joint arthroplasty, J. Biomed. Mater. Res. B., 107, 1587–1597, https://doi.org/10.1002/jbm.b.34251, 2019.
Lee, S. I., Lee, J. H., Park, S. Y., and Park, J. W.: Do bupivacaine, clindamycin, and gentamicin at their clinical concentrations enhance rocuronium-induced neuromuscular block?, Korean J. Anesthesiol., 64, 346, https://doi.org/10.4097/kjae.2013.64.4.346, 2013.
Martínez-Moreno, J., Merino, V., Nácher, A., Rodrigo, J. L., Climente, M., and Merino-Sanjuán, M.: Antibiotic-loaded bone cement as prophylaxis in total joint replacement, Orthop. Surg., 9, 331–341, https://doi.org/10.1111/os.12351, 2017.
Mikkelsen, M. K. D., Jørgensen, A. R., Kotla, N. G., Stilling, M., Damsgaard, M. B., Crocoll, C., Poborsky, M., Rasmussen, H. C., Henriksen, J. R., Hansen, A. E., and Bue, M.: CarboCell G/C offers high and prolonged concentrations of gentamicin and clindamycin in bone tissue following intraosseous injection, J. Bone Joint Infect., 10, 327–334, https://doi.org/10.5194/jbji-10-327-2025, 2025.
Minelli, E. B., Benini, A., Magnan, B., and Bartolozzi, P.: Release of gentamicin and vancomycin from temporary human hip spacers in two-stage revision of infected arthroplasty, J. Antimicrob. Chemoth., 53, 329–334, https://https://doi.org/10.1093/jac/dkh032, 2004.
Mouton, J. W., Theuretzbacher, U., Craig, W. A., Tulkens, P. M., Derendorf, H., and Cars, O.: Tissue concentrations: do we ever learn? J. Antimicrob. Chemoth., 61, 235–237, https://doi.org/10.1093/jac/dkm476, 2007.
Müller, M., Dela Peña, A., and Derendorf, H.: Issues in pharmacokinetics and pharmacodynamics of anti-infective agents: distribution in tissue, Antimicrob. Agents Ch., 48, 1441–1453, https://doi.org/10.1128/AAC.48.5.1441-1453.2004, 2004.
Olsen Kipp, J., Hanberg, P., Slater, J., Møller Nielsen, L., Storgaard Jakobsen, S., Stilling, M., and Bue, M.: Vancomycin bone and tissue concentrations following tibial intraosseous administration – evaluated in a porcine model, J. Bone Joint Infect., 6, 99–106, https://doi.org/10.5194/jbji-6-99-2021, 2021.
Pea, F.: Penetration of antibacterials into bone: what do we really need to know for optimal prophylaxis and treatment of bone and joint infections?, Clin. Pharmacokinet., 48, 125–127, https://doi.org/10.2165/00003088-200948020-00003, 2009.
Russo, M., Monnin, C., Zhang, Y. L., Montreuil, J., Tanzer, M., Avizonis, D., and Hart, A. A.: A novel method for quantification of cefazolin local tissue concentration in blood, fat, synovium, and bone marrow using liquid chromatography–mass spectrometry, J. Pharm. Biomed. Anal., 234, 115566, https://doi.org/10.1016/j.jpba.2023.115566, 2023.
Schwarz, E. M., McLaren, A. C., Sculco, T. P., Brause, B., Bostrom, M., and Kates, S. L.: Adjuvant antibiotic-loaded bone cement: concerns with current use and research to make it work, J. Orthop. Res., 39, 227–239, https://doi.org/10.1002/jor.24616, 2021.
Selby, N. M., Shaw, S., Woodier, N., Fluck, R. J., and Kolhe, N. V.: Gentamicin-associated acute kidney injury, QJM, 102, 873–880, https://doi.org/10.1093/qjmed/hcp143, 2009.
Steadman, W., Chapman, P. R., Schuetz, M., Schmutz, B., Trampuz, A., and Tetsworth, K.: Local antibiotic delivery options in prosthetic joint infection, Antibiotics, 12, 752, https://doi.org/10.3390/antibiotics12040752, 2023
Stolle, L. B.: Application of microdialysis to cancellous bone tissue for measurement of gentamicin levels, J. Antimicrob. Chemoth., 54, 263–265, https://doi.org/10.1093/jac/dkh291, 2004.
Vittrup, S., Jensen, L. K., Hartmann, K. T., Aalbæk, B., Hanberg, P., Slater, J., Hvistendahl, M. A., Stilling, M., Jørgensen, N. P., and Bue, M.: Rifampicin does not reduce moxifloxacin concentrations at the site of infection and may not improve treatment outcome of a one-stage exchange surgery protocol of implant-associated osteomyelitis lesions in a porcine model, APMIS, 132, apm.13371, https://doi.org/10.1111/apm.13371, 2023.
Zhao, Y., Prideaux, B., Baistrocchi, S., Sheppard, D. C., and Perlin, D. S.: Beyond tissue concentrations: antifungal penetration at the site of infection, Med. Mycol., 57, S161–S167, https://doi.org/10.1093/mmy/myy067, 2019.
Zimmerli, W. and Moser, C.: Pathogenesis and treatment concepts of orthopaedic biofilm infections, FEMS Immunol. Med. Mic., 65, 158–168, https://doi.org/10.1111/j.1574-695X.2012.00938.x, 2012.